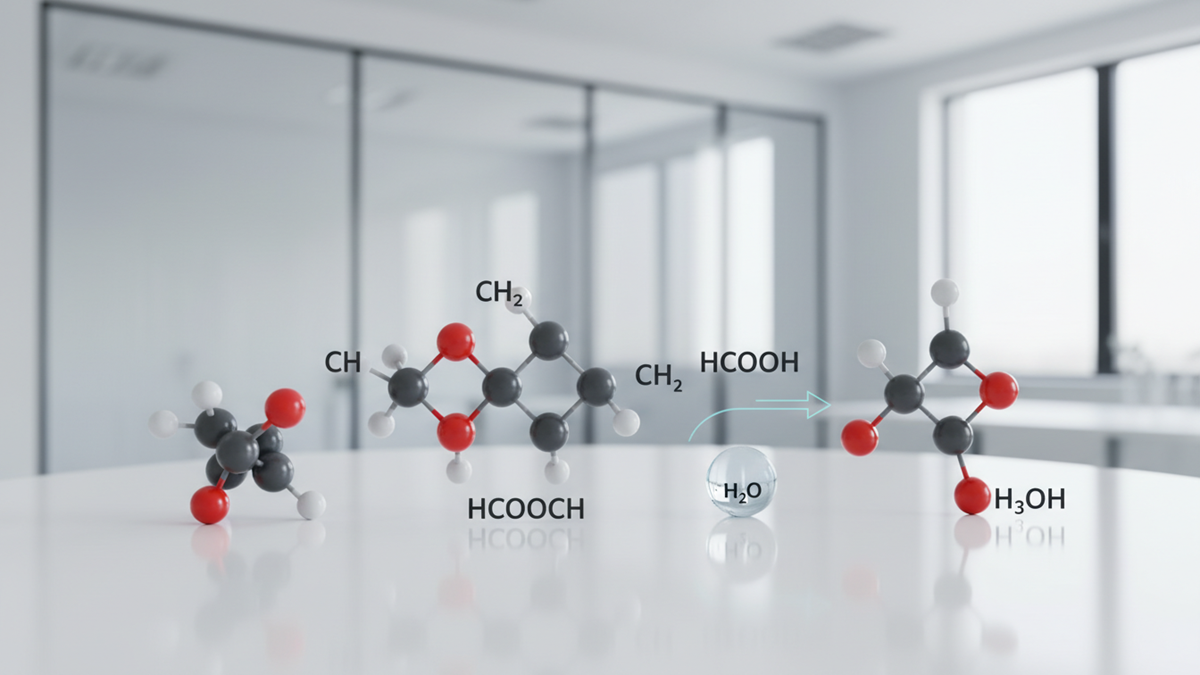
The Mysterious Molecule often appears in scientific discussions because its formula raises questions about how certain organic compounds behave under real conditions. Many people see the expression HCOOCH₃ + CH₂ + H₂O and assume it hides a complex idea, yet the core concept stays simple. It shows how carbon-based parts move, separate, and form new structures once the right environment appears.
Researchers in the United States follow compounds like this to study energy production, environmental changes, and industrial development. Each part of the formula gives them clues about how carbon-based molecules behave when heat, pressure, or moisture affect them. This article breaks the formula into plain language, offers clear examples, and shows why this so-called “Mysterious Molecule” continues to attract scientific attention.
What the “Mysterious Molecule” Really Means
The phrase “Mysterious Molecule” refers to a reaction that centers on a compound linked to esters, carbon chains, and small reactive groups. The formula does not point to one fixed product. Instead, it highlights how organic components may break apart, attach to new partners, or shift into different forms once heat, moisture, or catalysts appear. These movements help researchers map the hidden steps that drive many reactions in real systems.
Scientists rely on formulas like this to study how atoms move and settle into new arrangements. Each symbol in the formula marks a part of the process, from the behavior of the ester to the action of the carbon unit and the influence of water. When examined together, they show how complex reactions unfold inside laboratories, engines, and natural environments across the United States. This insight helps researchers understand how materials degrade, how fuels form, and how chemical pathways support modern technology.
Breaking the Formula Into Clear Pieces
HCOOCH₃: A Common Ester in Organic Chemistry
HCOOCH₃ represents methyl formate, an ester with a strong presence in chemical manufacturing. It carries a sweet smell and appears in various solvent blends. Producers rely on it due to its low cost and helpful reaction properties.
It also gives scientists clues about how esters break down or transform when exposed to heat, light, or certain catalysts.
CH₂: A Reactive Carbon Fragment
CH₂ stands for a single carbon group without full hydrogen saturation. Chemists treat it as a reactive unit inside larger reactions. It explains how carbon chains grow in fuels, plastics, and synthetic materials.
Its behavior often determines whether a reaction moves fast, slow, or fails altogether.
H₂O: Water as a Reaction Driver
Water shapes chemical outcomes more than most people realize. It can support or limit a reaction based on temperature and environment.
In this formula, water helps stabilize or separate molecules, giving the reaction new direction.
How These Parts Work Together
The three components highlight a broader pattern in organic chemistry. The ester may shift when exposed to a CH₂ fragment and water, which means new products can form. These interactions help researchers study energy pathways, environmental breakdown, and industrial processing.
U.S. laboratories often track similar reactions to understand biofuel production, petroleum behavior, or environmental cleanup methods.
Why Scientists Call It “Mysterious”
The molecule gained attention due to its unusual combination. It does not point to one final product. Instead, it guides researchers to explore how carbon groups attach, detach, or reorganize.
This mystery keeps chemists focused on discovering new reaction steps that may lead to efficient energy production or eco-friendly materials.
Real-World Uses and Relevance in the United States
1. Fuel Technology
U.S. energy labs study ester-based reactions to improve fuel stability. These reactions help researchers see how fuel components break down inside engines.
2. Environmental Research
The combination of esters, carbon fragments, and water appears in pollution studies. Scientists track how airborne esters behave when sunlight or humidity changes.
3. Industrial Manufacturing
Methyl formate plays a role in adhesives, foams, and solvent systems. Understanding how it interacts with other carbon parts helps industries produce safer, cleaner materials.
How Scientists Analyze This Molecule
Researchers rely on tools such as:
• Mass spectrometry
• Infrared analysis
• Controlled reaction chambers
These tools help them understand how fast the reaction moves, what products appear, and how conditions such as heat, pressure, or moisture affect outcomes.
What Makes This Study Valuable
The molecule teaches scientists how organic compounds shift under different conditions. This knowledge leads to stronger materials, cleaner fuels, and reduced environmental impact.
Its flexibility keeps researchers curious and encourages deeper exploration in schools, labs, and industrial settings.
Conclusion
The “Mysterious Molecule” may look unusual at first, yet its meaning becomes clear once each part of the formula is separated and explained. The ester, the reactive carbon piece, and the influence of water show how organic molecules follow steady pathways once the right conditions appear. These reactions give scientists a clearer view of how to develop new materials, cleaner fuels, and more efficient industrial systems.
Researchers in the United States focus on reactions like this since they uncover the ways carbon structures shift under real environmental conditions. The information gained from these reactions supports progress in manufacturing, environmental science, and energy development. The molecule remains mysterious only until the science behind it is understood.
FAQs
1. Why is this molecule considered mysterious?
It suggests multiple reaction pathways instead of one simple answer, which turns it into a puzzle for chemists.
2. Does methyl formate appear in U.S. industries?
Yes. Manufacturers use it in foams, solvents, and chemical processing.
3. Does CH₂ occur alone in the real world?
CH₂ usually appears inside larger reactions. It rarely exists in isolation for long.
4. Why does water matter in this formula?
Water influences reaction steps, speeds, and product stability.
5. Does this molecule relate to fuel production?
Researchers often study similar reactions to understand fuel breakdown and improvement.
6. Can students understand this reaction easily?
Yes. Once the formula is broken into parts, the concept becomes simple.
7. What industries benefit from this study?
Energy, manufacturing, environmental research, and chemical production.